40 mo diagram for hcl
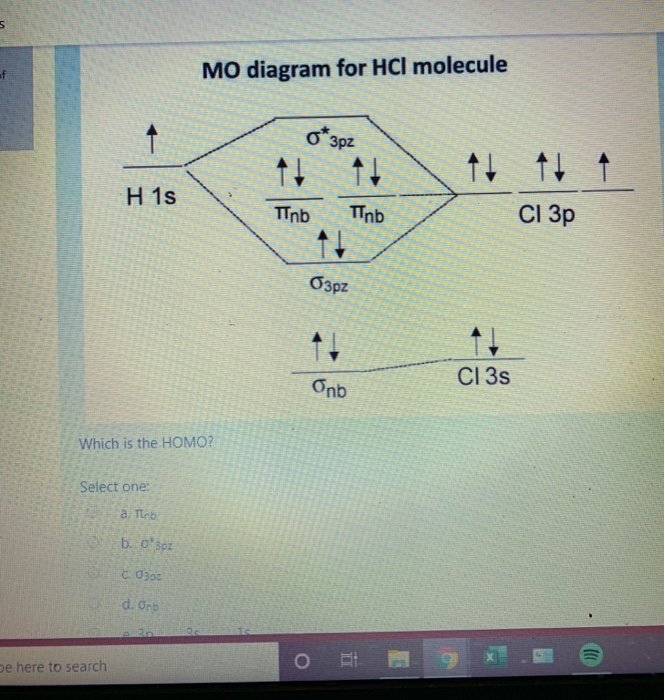
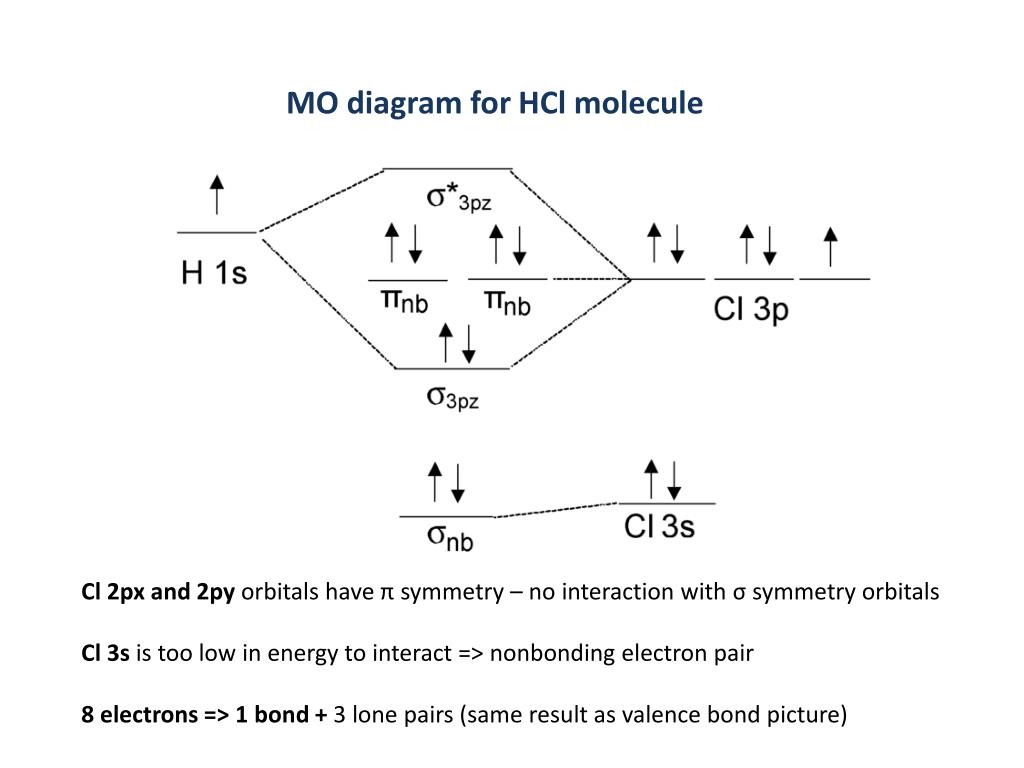
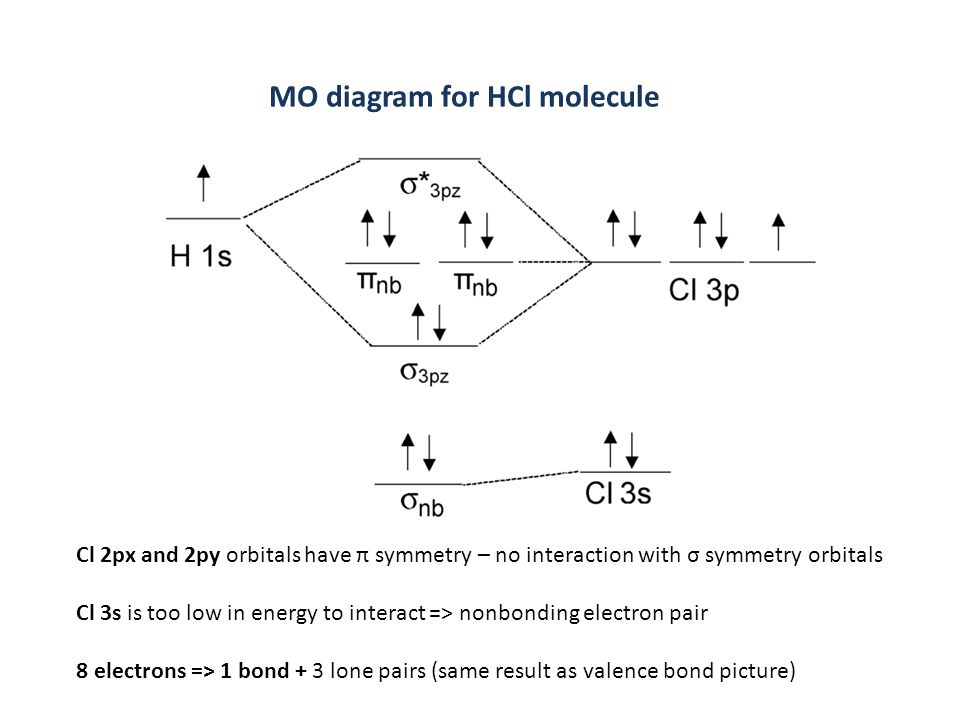
Here is a useful MO diagram of HCL found on the internet: The Cl electrons residing up to 3s orbital (1s, 2s, 2px,2py,2pz,3s) are largely stabilized than H ... First, identify the number of valence electrons. The number of valence electron per element is based on the group number. Next, fill the molecular orbitals ( ...
How are the 3p orbitals of chlorine lower in energy than the 1s orbital of hydrogen? MO diagram of HCl. Share. Share a link to this question. Copy link
Mo diagram for hcl
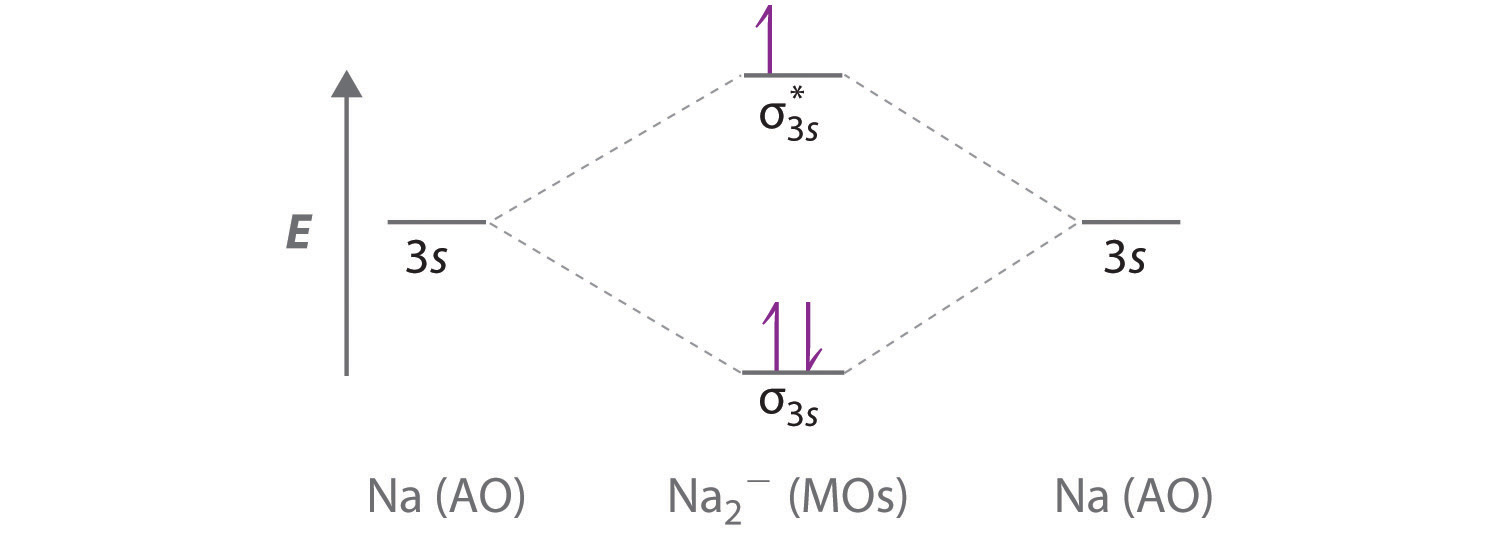
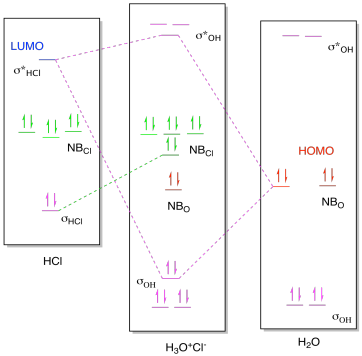
Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal.Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23 Na. The free metal does not occur in nature, and must be prepared from compounds. To use molecular orbital theory to predict bond order ... Figure 11.5.14: Molecular Orbital Energy-Level Diagram for HCl. A heteronuclear diatomic molecule is composed of two atoms of two different elements. Examples include CO, HCl, and NO. Dihydrogen[edit]. H2 ...
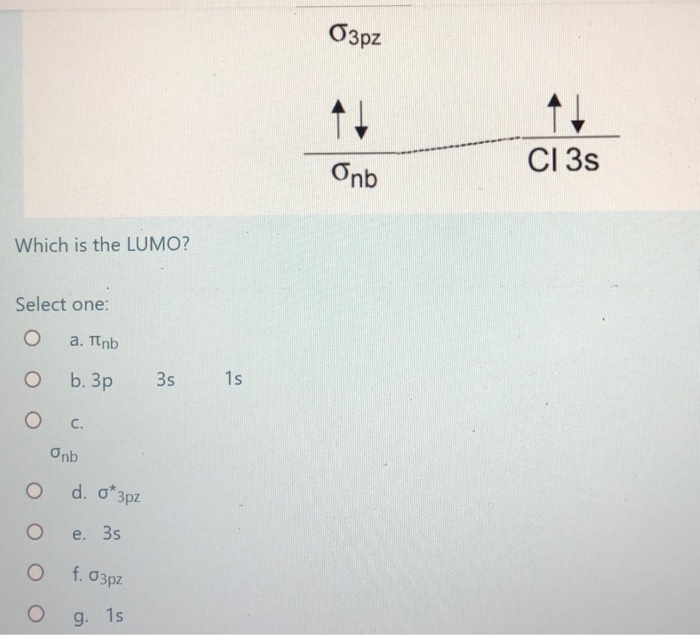
Mo diagram for hcl. Therefore, the HCL molecule has 8 pairs (1s, 2s, 2px,2py,2pz,3s,3px and 3py) of non-bonding (nb) electrons and one bonding (sigma) orbital ... For notes click here-https://trickychemistrysuman.blogspot.com/2020/03/mo-diagram-of-hcl.html. Take glucose and HCl. Both are polar molecules that have dipole-dipole interactions, and have hydrogens with partial positive charges. However, HCl separates (dissociates) into ions in solution, whereas individual glucose molecules stay together and are each surrounded by water molecules (they dissolve). 6 Sept 2020 ... DescriptionHCl mo diagram.png. English: MO energy level diagram for HCl. Source, Own work. Author, Tem5psu ...
This lecture clearly explains the molecular orbital diagram of heteronuclear diatomic molecule (HF & HCl). This will help students of H.S., ... Problem 54 Hard Difficulty. Draw an MO energy diagram for HCl. Predict the bond order and make a sketch of the lowest energy bonding molecular orbital. A heteronuclear diatomic molecule is composed of two atoms of two different elements. Examples include CO, HCl, and NO. Dihydrogen[edit]. H2 ... To use molecular orbital theory to predict bond order ... Figure 11.5.14: Molecular Orbital Energy-Level Diagram for HCl.
Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal.Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23 Na. The free metal does not occur in nature, and must be prepared from compounds.
Solved Assume That The Mo Diagram Of Hcl Is Analogous To That Of Hf A Comment On The Main Differences Between The Diagrams For Hf And For Hcl B What Basis Set Of

Hydrogen Chloride Hcl Molecule Lewis Dot Cross Electronic Diagram Covalent Bonds Ball Stick Space Filling 3d Models Boiling Point Melting Point Doc Brown S Chemistry Revision Notes
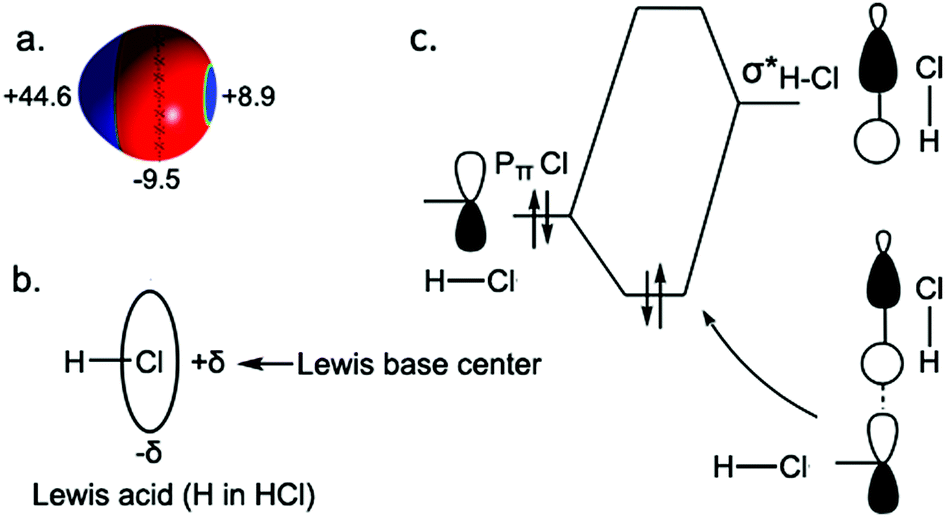
Emergence Of Novel Hydrogen Chlorides Under High Pressure Physical Chemistry Chemical Physics Rsc Publishing Doi 10 1039 C6cp08708f

Give Orbital Diagram Of The Following A Magnesium Chloride B Nitrogen C Methane D Hydrogen Chloride Flash Education

Give Orbital Diagram Of The Following A Magnesium Chloride B Nitrogen C Methane D Hydrogen Chloride Flash Education

Give Orbital Diagram Of The Following A Magnesium Chloride B Nitrogen C Methane D Hydrogen Chloride Flash Education



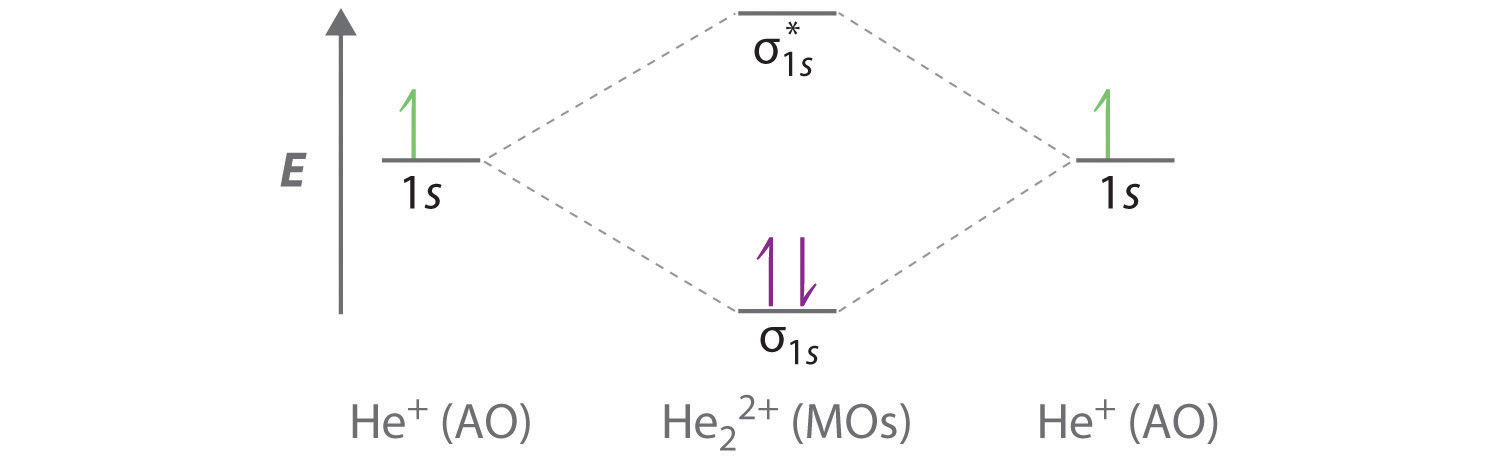











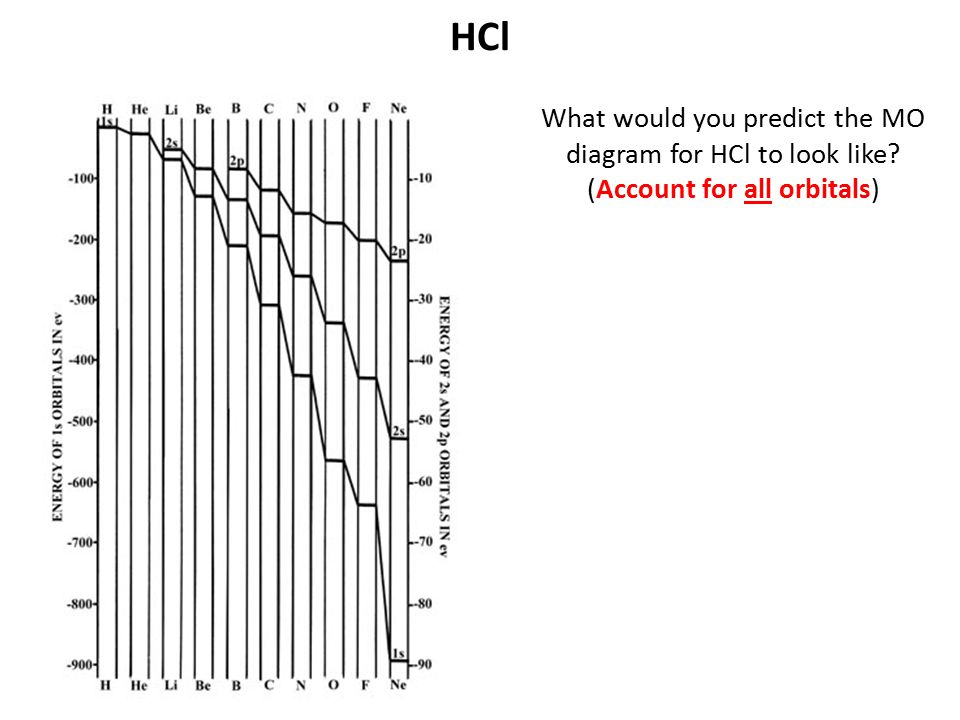



0 Response to "40 mo diagram for hcl"
Post a Comment